Buxton Heath







Dung Fungus Sp (Cheilymenia Sp)
Steve's thoughts on identifying this one
On to the little orange jobbies on the cow poop, under the dissecting scope the long pale lashes were very visible and rather dramatic,
there was a complete lack of the star shaped cluster of hairs at the base of each of the long hairs, which rules out Cheilymenia stercoria.
I can struggle with some of the similarities amongst the Cheilymenias so tend towards looking at microscopy sooner than I would with some other groups.
This paid off this time with the spores being long ovals with a very distinct DeBary bubble in the middle, looking like a large round oil drop in the middle,
this according to Peter Thompson points to Cheilymenia coprinaria rather than Cheilymenia fimicola as does the slightly narrower shape of the spores.
This view was supported by Fungi of temperate Europe, everything pointing nicely to Cheilymenia coprinaria, so a quick check on FRDBI
(the fungal records database of Britain and Ireland), where all our records ultimately go, which says that Cheilymenia coprinaria is a synonym of Cheilymenia fimicola.
A paper written in 1958 suggests that DeBary bubbles are actually a reaction to the chemical and squashing under a cover slip of the microscopy process rather than
an identification feature of the fungi so all in all safer to go with Cheilymenia fimicola. 15 minutes ID, 25 minutes research to resolve the findings.
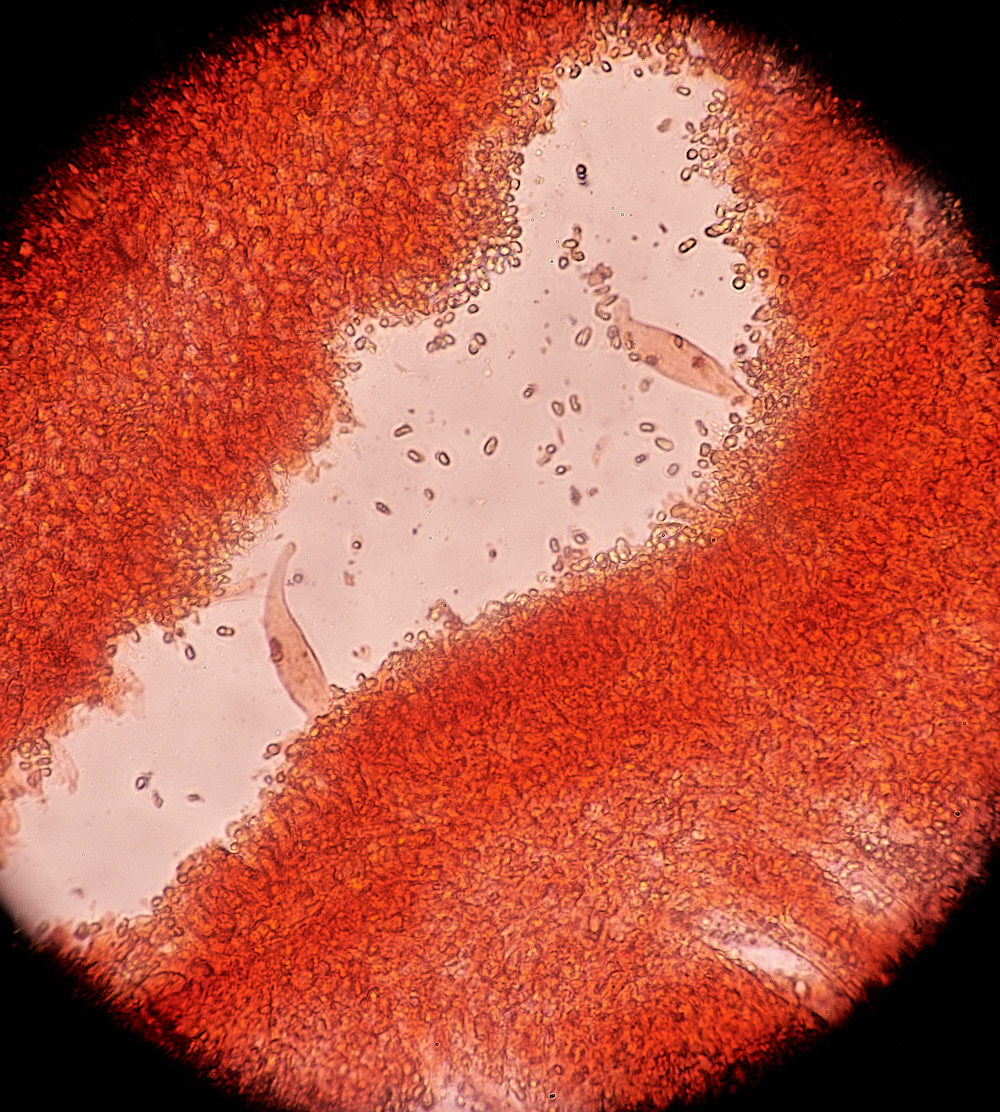
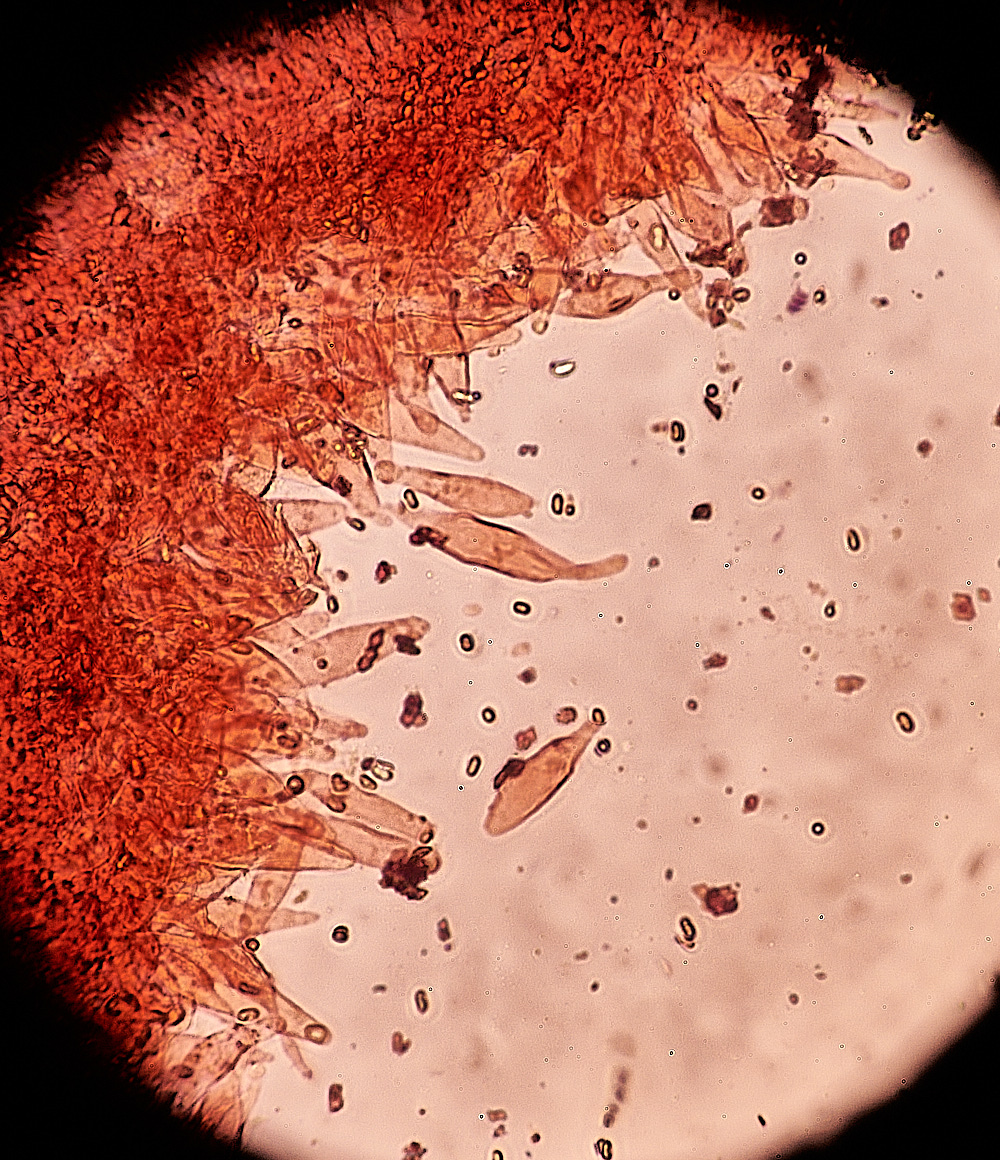
The Black-edged Gills of Blackedge Bonnet (Mycena pelianthina)
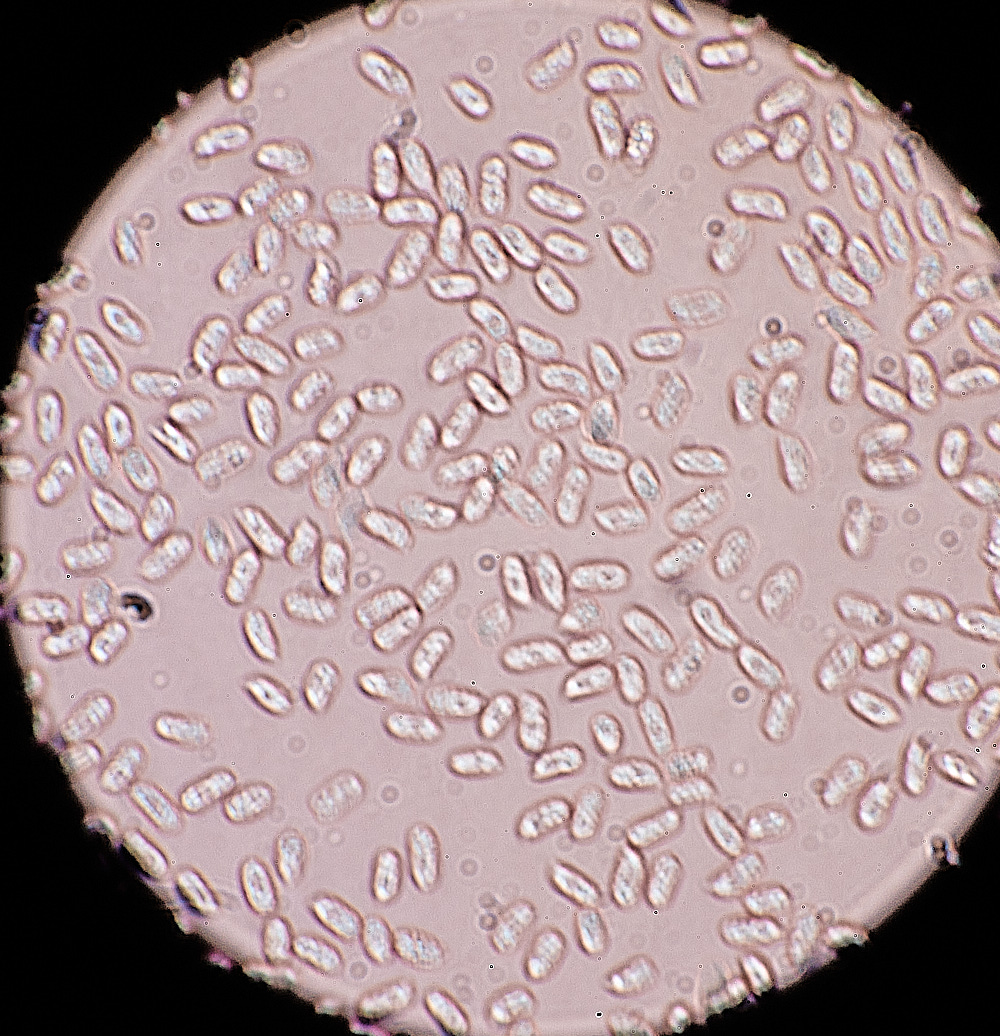
3.5 x 8.0 µm
Cheilocystidia found on the gill edge. Mycena pelianthina)


Nail Fungus (Poronia punctacta)
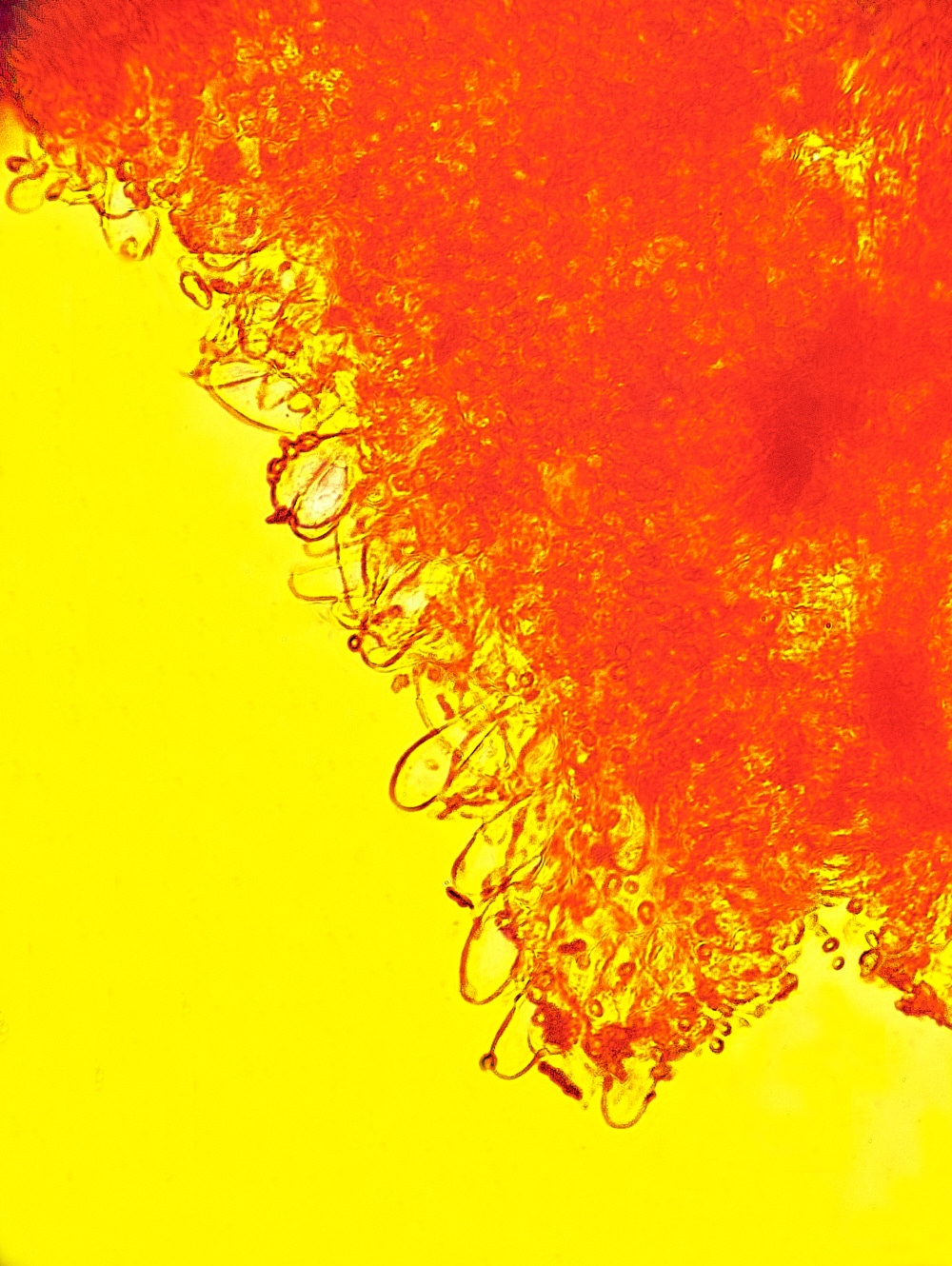
Common Bonnet (Mycena galericulata)
Plain Earthtongue (Geoglossum umbratile)
Steve's delightful exposition of how he reached this conclusion.
To finish off before lunch with a nice easy quick one it was on to the Earth tongue (another one that appeared just after you left Lorraine).
No point going to Kibby as he doesn't do ascomycetes and neither does Funga Nordica. So in the absence of forking out £130 for
B & K Fungi of Switzerland, Volume 1: Ascomycetes, we are left with Peter Thompson and Fungi of temperate Europe.
Starting with Peter Thompson as he does nice drawings of spores etc. there are four to choose from. Geoglossum have septate spores,
one of the options has 6-9 septa, one has 13-15 septa, one has 5-7 septa and one has 3-7, nice and easy just count the septa I thought.
Unfortunately the spores have no septa when young and develop the septa as they get older so we are trying to find spores that have been
released from the asci and are mature enough to have developed their full quota of septa. Scraping a very small part of the surface of the
fruiting body and spreading it on a slide, I started out with Meltzer's as the asci react by bluing but then you can't see the spores as there
is too much blue to see through, try again with plain water as the colour of the spores is important, not worth doing as without stain
nothing stood out, try again this time with plaq search, much better and there are anything between 0 and 7 septa, which fits nicely
with G. glutinosum, except as implied by the name it should have a glutinous layer and it didn't, oh and the spores are the wrong size.
So lets try G. Cookeanum, number of Septa is right but shape of fruiting body is wrong and our spores have oil drops and G. Cookeanum doesn't.
By this time hunger is kicking in so break for lunch. Return refreshed and look at Fungi of temperate Europe, lots more to choose from but no helpful drawings.
Time to accept that we are going to have to use a key so go for Geoglossaceae of Britain - Brian Spooner, realise that we are going to have to find some
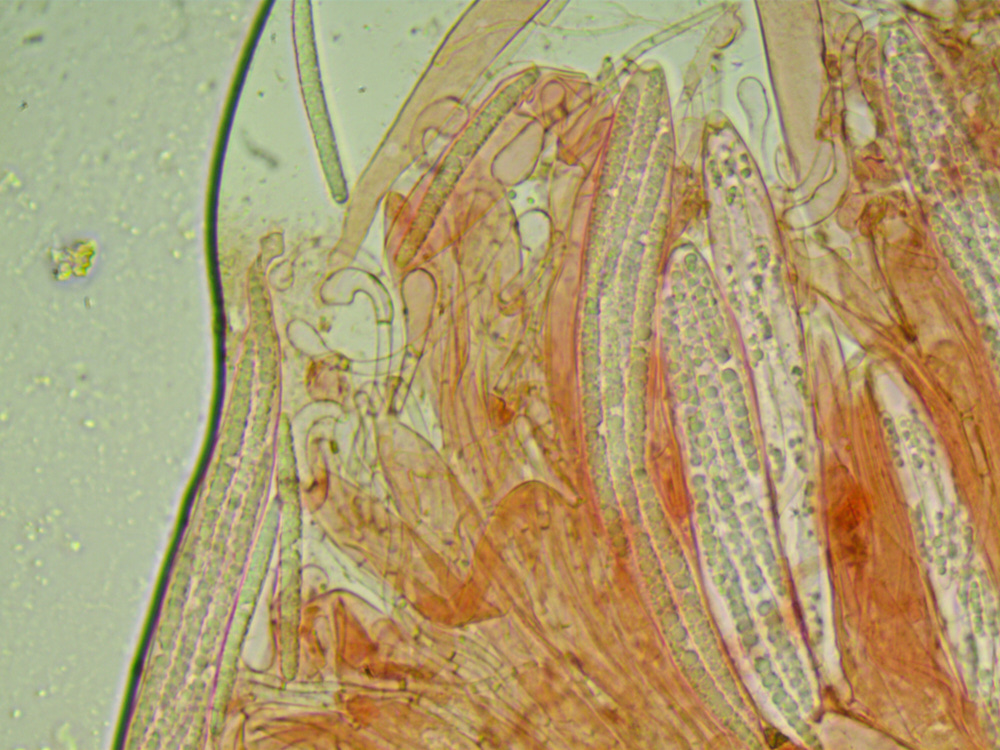
paraphyses amongst the asci, (paraphyses are, to use an obscure music reference, the bits between the bits). So another slide prep this time with
cotton blue as this is supposed to be good with asccomycetes, no paraphyses found, another slide prep, this time with my old favourite plaq search,
actually found one and managed to count the septa, (some species have paraphyses with septa and the number and spacing of the septa feeds into the ID)
but our septa have droopy heads which isn't mentioned in the key. Every attempt at the key takes us to G. glutinosum, but it isn't glutinous.
Another slide prep with Cong red and loads of nice paraphyses with drooping tips. Moving on to a document with key that seems more aimed
at our knowledge level, Geoglossaceae - Earth Tongues Beginners Guide - Irene Ridge. First up it is written in a nice friendly way and joy of joys
it has nice drawings and there we find spores of the right size with up to seven septa, paraphyses with the drooping tips Geoglossum umbratile - Plain Earthtongue.
Going back and reading Fungi of temperate Europe again it very clearly states that Geoglossum umbratile has paraphyses with drooping tips. Time wise dare I admit to
the fact that I spent well over 4 hours on this one fungi.
Photos of the spores and the paraphyses.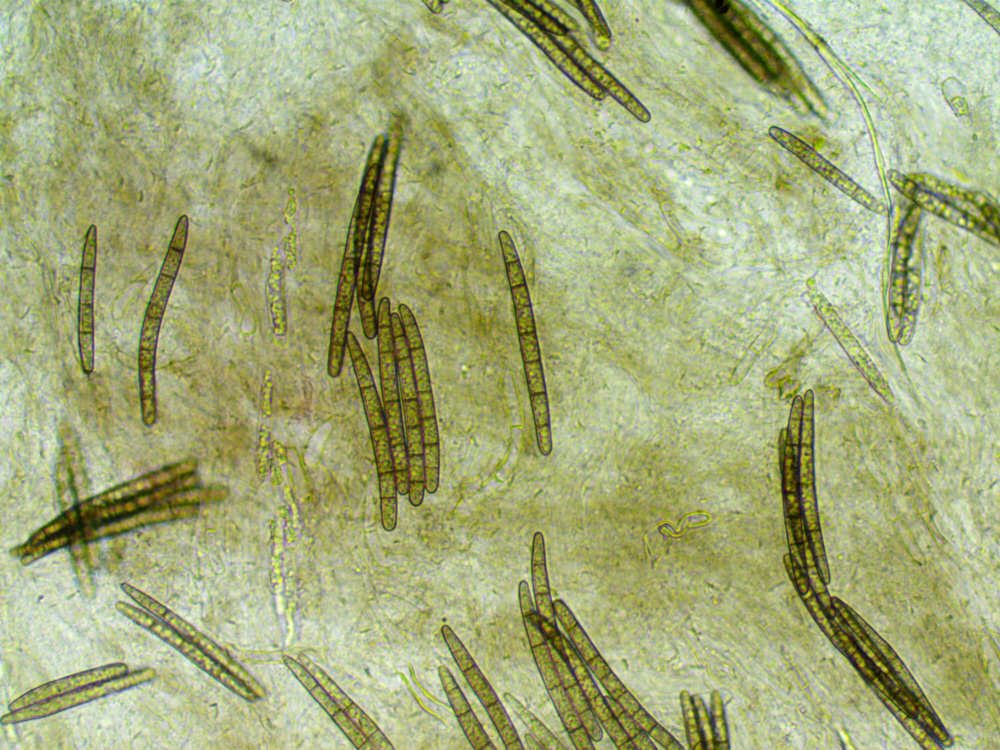


Fairy Fingers (Clavaria fragilis) perhaps?
Microscopically this turned out to be
Pointed Club (Clavaria acuta)
Thanks to Steve Judd for id & the thinking below.
Then we attacked the rather nice white Clavaria (sadly these appeared just after you left Lorraine) that were growing in the grass near the end,
we were fairly sure that this would be fairly easy as there really aren't that many white Clavaria. Using Kibby vol 1
we worked out that the growth habit indicated C. crosslandii or C. acuta the shape pointed towards C. acuta or C. fragilis.
A fine sliver taken with a razor blade to get to the spores, stained with plaq search. C. fragilis has heavily ornamented spores and these were smooth, C. crosslandii
has spores half the size of those of C. acuta so it was very easy to decide that we have Clavaria acuta - Pointed Club. 10-15 minutes

Trametes Sp?

The Blusher (Amanita rubescens)
Small Stagshorn (Calocera cornea)
Russula Sp

Stinkhorn Egg (Phallus impudicus)

Heath Star-moss (Campylopus introflexus) & Bank Haircap (Polytrichum formosum)

Fly Agaric (Amanita muscaria)


Broom Fork-moss (Dicranum scoparium)


Crimped Gill (Plicatura crispa)

Bank Haircap (Polytrichum formosum)
Pointed Spear-moss (Calliergonella cuspidata)
